| Dr William Small, Lead Applications Scientist, Croda Europe |
Introduction
The efficacy of topical formulations that contain active ingredients depends not only on the potency of the active ingredient, but also on other ingredients present in the formulation. The choice of formulation type (eg w/o or o/w emulsion, gel, ointment), emollients, penetration enhancers and even the emulsifier can significantly affect both the speed of delivery and location within the skin of the active ingredient, both of which will impact on the efficacy of the product. This article will discuss some of the ingredient choices and formulation strategies that can be used in the early development stages or re-design of topical formulations.
Background
The skin is a common and popular route of administration for both cosmetic (eg skin lighteners and tanners) and pharmaceutical (eg steroid) active ingredients; however it is also a very challenging route. The structural complexity of the skin means that this route is only really suitable for active ingredients with quite a narrow profile in terms of molecular weight and lipophilicity. Even then, it can be difficult to achieve good bioavailability – a sufficient quantity of active, delivered to the right location in the skin within a suitable time frame so as to be efficacious.
Controlling the delivery rate – creating a ‘push’
From the moment that the formulation is applied to the skin, there is a finite amount of time within which the active ingredient has an opportunity to leave the formulation and penetrate the upper layers of the skin. Beyond this time the active is likely to have crystallised or been left stranded on the surface of the skin due to the evaporation of aqueous or alcoholic ingredients or the absorption of other ingredients into the skin. One way to use this window of permeating opportunity effectively is to make sure that the active ingredient is being ‘pushed out’ of the formulation and into the skin, which can be done in a number of ways. One method is to simply increase the concentration of the active ingredient in the formulation, which will result in more active ingredient getting into the skin. A more elegant method is to formulate the active at or close to its maximum solubility, at high thermodynamic activity. This creates a driving force for the active ingredient to leave the formulation and penetrate the skin. We can illustrate this in the following example.
Three model formulations with salicylic acid have been prepared. The concentration of active ingredient is the same in each formulation, but different oil phases have been used, in which the solubility of salicylic acid varies. In one formulation, the concentration is close to the maximum solubility and so the thermodynamic activity is high. In another formulation, the maximum solubility is somewhat higher than the concentration and so the thermodynamic activity is a little lower than the first formulation. In the third formulation, the maximum solubility of salicylic acid is higher again and thus the thermodynamic activity is even lower. As shown in Figure 1, it is the thermodynamic activity that influences the delivery of the active ingredient – higher thermodynamic activity results in higher flux. This demonstrates the importance of considering the ‘inactive’ ingredients when formulating – the effect that simply switching from one oil phase to another can have on delivery of the active is significant.
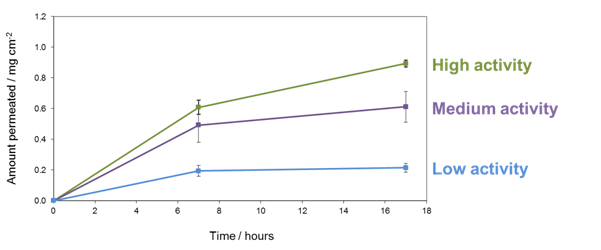
Figure 1: Permeation of salicylic acid through PDMS membranes. Concentration equivalent in each formulation, but different oil phases means that the thermodynamic activity is different in each formulation
Controlling the delivery rate – creating a ‘pull’
The formulating strategy to help ‘push’ the active ingredient out of the formulation has been discussed; however that is only tackling one part of a complex problem. It is generally acknowledged that active ingredients with a log P (partition coefficient) of between 1-3 are most suited to topical delivery, and once these have been ‘pushed’ from the formulation, they should penetrate the skin relatively easily. However, actives outside this range that are hydrophilic (log P<1) or very lipophilic (log P>3) do not find it as easy to penetrate the skin due to the alternating aqueous/non-aqueous regions of the intercellular lipids and hydrophilic domains. For these actives it can be useful to adopt a ‘pull’ strategy, where an excipient in which the active ingredient is soluble acts as a carrier vehicle, pulling the active into the skin. To do this, the excipient must both solubilise the active and show good solubility in the skin as well, acting as a penetration enhancer. Dimethyl isosorbide (DMI) is an example of an excipient that excels in this role.
In our next example two formulations were prepared with a model hydrophilic active, propagermanium. The formulations were simple creams, with one formulation including 10% dimethyl isosorbide and the control formulation replacing DMI with water. Permeation studies through porcine skin were conducted and the amount of active in the different compartments of the skin was measured after 20 hours. Figure 2 shows that the formulation containing 10% dimethyl isosorbide resulted in more active ingredient being located in the skin (after tape strips were removed) than the control formulation. Interestingly, the active was confined to the skin and tape strips and had not entered the receptor fluid. This suggests that DMI might be suitable for delivering actives into the skin without the actives entering systemic circulation.
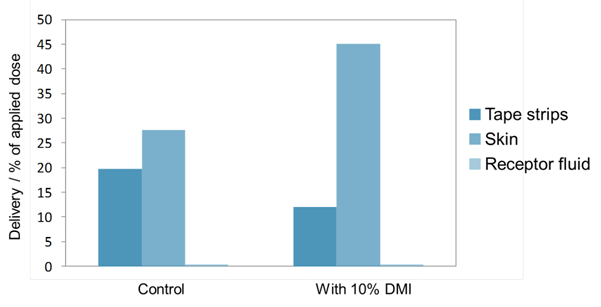
Figure 2: Permeation studies of Propagermanium through porcine skin, from two formulations with and without 10% dimethyl isosorbide (DMI)
Targeted delivery – hitting the right layer in the skin
We have looked briefly at controlling the rate of delivery and at delivering ‘challenging’ actives into the skin. But there is also something we can do as formulators to help control the location of the active ingredient in the skin. We can use liquid crystal-forming emulsifiers, ie surfactants that form liquid crystalline phases not just on evaporation of the aqueous phase, but in the formulation itself. There are two types of liquid crystalline structure that we have formed, the so-called oleosome and hydrosome structures. Oleosomes are liquid crystal regions that form around the oil drops in an emulsion, shown in Figure 3a, and appear under optical microscopy as ‘Maltese crosses’. These regions are thought to be made up of multiple surfactant bilayers that encapsulate the oil drops, providing stability to coalescence as well as targeted delivery for some active ingredients. Hydrosomes are liquid crystal regions that form throughout the aqueous regions of an emulsion. They can also be visualised under optical microscopy (Figure 3b), and can also be identified through rheology measurements as they give the formulation a unique rheological profile due to the lamellar phases that form in the continuous phase of the emulsion. Formulations containing hydrosome liquid crystals also have an aesthetically pleasing light feel, as the lamellar layers slide over each other under shear.
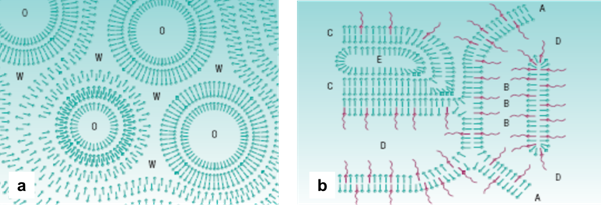
Figure 3: Oleosome (a) and hydrosome (b) liquid crystal structures. O = oil, W = water, A = hydrophobic part, B = entrapped water, C = hydrophilic part, D = external water phase
The effect of these two emulsifier systems on the delivery of lipophilic and hydrophilic active ingredients is shown in the following two examples. In Figure 4, the amount of lipophilic active ingredient (octadecenedioic acid) recovered from different strata of porcine skin samples is shown following treatment with two formulations. One formulation contains an oleosome emulsifier system (Brij™ S2/Brij S721, Croda), the other contains a hydrosome emulsifier system (Arlacel™ 2121, Croda). The data shows that more active ingredient is recovered deeper down, in the receptor fluid, from the hydrosome system than from the oleosome system. However more active ingredient is recovered in the skin itself from the oleosome system. This is just one example but is something that has been observed for multiple lipophilic actives.
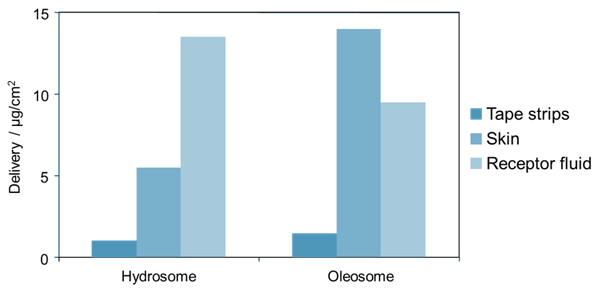
Figure 4: Permeation studies of a lipophilic active ingredient, octadecenedioic acid, through porcine skin. The formulations are identical, with the exception of the emulsifier used – one contains an oleosome-forming emulsifier system, the other a hydrosome-forming emulsifier system
For a hydrophilic active ingredient, propagermanium, an entirely different delivery profile is observed. Figure 5 presents the data from a study comparing the recovery of propagermanium from the different strata in porcine skin, following application with an oleosome- and a hydrosome-containing formulation. As the data shows, the amount of active present in the skin from the oleosome formulation is much lower than from the hydrosome formulation, while the receptor fluid from each study shows almost no active has permeated that far.
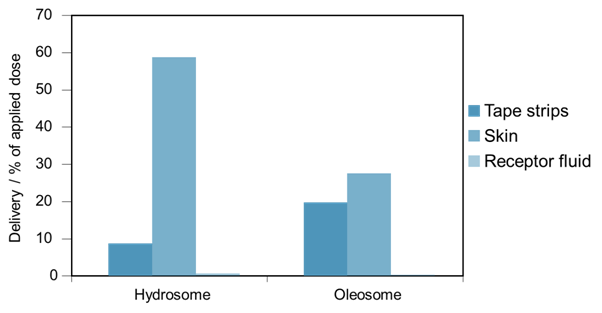
Figure 5: Permeation studies of a hydrophilic active ingredient, propagermanium, through porcine skin. The formulations are identical, with the exception of the emulsifier used – one contains an oleosome-forming emulsifier system, the other a hydrosome-forming emulsifier system
So why should the delivery of an active ingredient differ based on the emulsifier system used? For the lipophilic active ingredient contained in the oil phase of the emulsion, we suggest that the surrounding oleosome structure acts as a barrier to the active diffusing out of the formulation, slowing down its permeation. This results in more active becoming located within the skin. From the hydrosome formulation however, we believe that the increased hydration of the skin that is associated with hydrosome-containing formulations results in enhanced permeation of the active ingredient, resulting in faster transport through the skin and the active being mainly located in the receptor fluid in our studies. For the hydrophilic active, we believe that the hydrosome formulation helps to keep the active ingredient in solution for longer due to reduced evaporation, allowing it more time to diffuse in to the skin. However evaporation from the oleosome formulation means that the active is left stranded on the surface, and less able to diffuse in to the skin.
Summary
This article has described some of the ways in which the delivery of an active ingredient, in terms of its rate of permeation and location within the skin, can be controlled through formulation design. Every ingredient in the formulation has the potential to have some effect on the delivery of an active ingredient, either through its effect on the solubility of the active, its interaction with the skin, or on the formulation as it undergoes various phase changes on the skin. It is therefore important to consider the role of every ingredient in the formulation to optimise delivery of the active ingredient.


